About Erleada
Mechanism of action
ERLEADA® was designed to selectively and irreversibly bind to the androgen receptor (AR) and thus inhibit AR signaling at various locations:[1]
Watch this 1 minute video to discover the mode of action of Erleada.
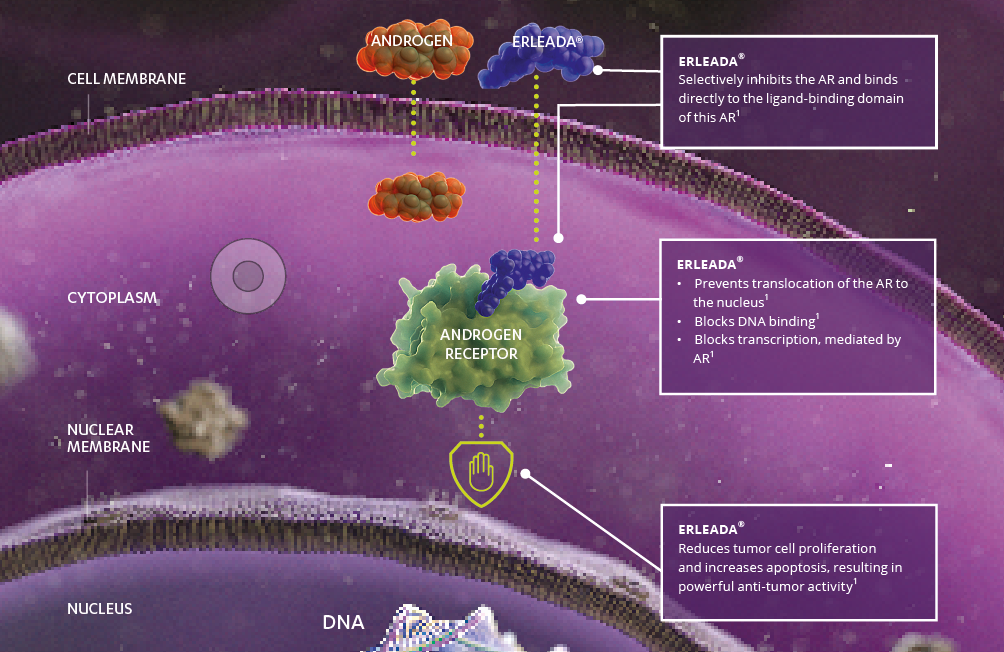
ERLEADA® has no agonistic effect on the AR.
Dosage & administration
- The recommended dose of ERLEADA® is 240 mg (four 60 mg tablets) as a single daily dose, with or without food[2]
- In patients who have not been surgically castrated, chemical castration with a gonadotropin-releasing hormone analog (GnRHa) should be continued during treatment[2]
ERLEADA® was designed to selectively and irreversibly bind to the androgen receptor (AR) and thus inhibit AR signaling at various locations:1

4 tablets x 60 mg once
daily oral dose

With or without food

Do not crush or break
the tablets
Forgot a dose?
- If a dose is missed[2], it should be taken as soon as possible on the same day, with the normal schedule resuming on the following day.
- Extra tablets should not be taken to make up the missed dose.
You can consult the summary of the product characteristics of Erleada:
Packaging

- The daily recommended dose of Erleada is 240 mg (= 4 tablets of 60 mg per day)[1]
- A box of Erleada contains 4 blisters of each 28 tablets (112 tablets in total)
Erleada | Janssen product code | CNK code | Registration nr | Price ex-factory excl. VAT |
112 x 60 mg tab | 417715 | 3889-607 | EU/1/18/1342/001 | € 2925,56 |
Contraindications
The use of Erleada® is contraindicated in the following cases[^1]:

In the event of hypersensitivity to any of the ingredients in this medicine°

In women who are pregnant or could become pregnant
Side effects
Summary of the undesirable effects
The most common side effects are fatigue (26%), rash (26% all grades together and 6% grade 3 or 4), hypertension (22%), hot flushes (18%), arthralgia (17%), diarrhea (16%), falls (13%) and weight loss (13%). Other significant side effects include fractures (11%) and hypothyroidism (8%).[1]
Undesirable effects identified in clinical studies
System organ class | Adverse reaction & frequency |
|---|---|
System organ class Endocrine disorders | Adverse reaction & frequency Common: hypothyroidisma |
System organ class Metabolism and nutrition disorders | Adverse reaction & frequency Very common: decreased appetite |
System organ class Nervous system disorders | Adverse reaction & frequency Common: dysgeusia |
System organ class Cardiac disorders | Adverse reaction & frequency Not known: QT prolongation |
System organ class Vascular disorders | Adverse reaction & frequency Very common: hot flush, hypertension |
System organ class Gastrointestinal disorders | Adverse reaction & frequency Very common: diarrhoea |
System organ class Skin and subcutaneous tissue disorders | Adverse reaction & frequency Very common: skin rashd
|
System organ class Musculoskeletal and connective tissue disorders | Adverse reaction & frequency Very common: fracturef, arthralgia
|
System organ class General disorders and administration site conditions | Adverse reaction & frequency Very common: fatigue |
System organ class Investigations | Adverse reaction & frequency Very common: weight decreased |
System organ class Injury, poisoning and procedural complications | Adverse reaction & frequency Very common: fall |
a Includes hypothyroidism, blood thyroid stimulating hormone increased, thyroxine decreased, autoimmune thyroiditis, thyroxine free decreased, tri-iodothyronine decreased
b Includes tongue biting
c Includes angina pectoris, angina unstable, myocardial infarction, acute myocardial infarction, coronary artery occlusion, coronary artery stenosis, acute coronary syndrome, arteriosclerosis coronary artery, cardiac stress test abnormal, troponin increased, myocardial ischaemia
d See “Skin rash” under “Description of selected adverse reactions”
e Post-marketing adverse reaction
f Includes rib fracture, lumbar vertebral fracture, spinal compression fracture, spinal fracture, foot fracture, hip fracture, humerus fracture, thoracic vertebral fracture, upper limb fracture, fractured sacrum, hand fracture, pubis fracture, acetabulum fracture, ankle fracture, compression fracture, costal cartilage fracture, facial bones fracture, lower limb fracture, osteoporotic fracture, wrist fracture, avulsion fracture, fibula fracture, fractured coccyx, pelvic fracture, radius fracture, sternal fracture, stress fracture, traumatic fracture, cervical vertebral fracture, femoral neck fracture, tibia fracture.
SmPC
Learn more on Erleada (apalutamide) via this dynamic, interactive video on:
- The mode of action
- The dosing and administration
- The registration trials SPARTAN (nmCRPC) and TITAN (mHSPC)

GnRH = gonadotropin-releasing hormone
AR = androgen receptor
DNA = deoxyribonucleic acid
° The active substance in this medicine is apalutamide. Each film-coated tablet contains 60 mg of apalutamide. The other ingredients in the tablet core of this medicine are colloidal anhydrous silica, croscarmellose sodium, hypromellose acetate succinate, magnesium stearate, microcrystalline cellulose and silicified microcrystalline cellulose. The film coating contains black iron oxide (E172), yellow iron oxide (E172), macrogol, polyvinyl alcohol (partially hydrolyzed), talc and titanium dioxide (E171).
Erleada is indicated:1
- for the treatment of metastatic hormone-sensitive prostate cancer (mHSPC) in adult men, in combination with androgen deprivation therapy (ADT)
- for the treatment of non-metastatic castration-resistant prostate cancer (nmCRPC) in adult men with a high risk of developing metastatic disease in combination with ADT
Erleada is reimbursed as of the 1st of September 2021 in Belgium. Price ex-factory excl. VAT: €2952,56
Erleada is reimbursed as of the 1st of July 2020 in Luxembourg. Public Price: € 3114,65
▼ This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions. See section “Undesirable effects” for how to report adverse reactions.